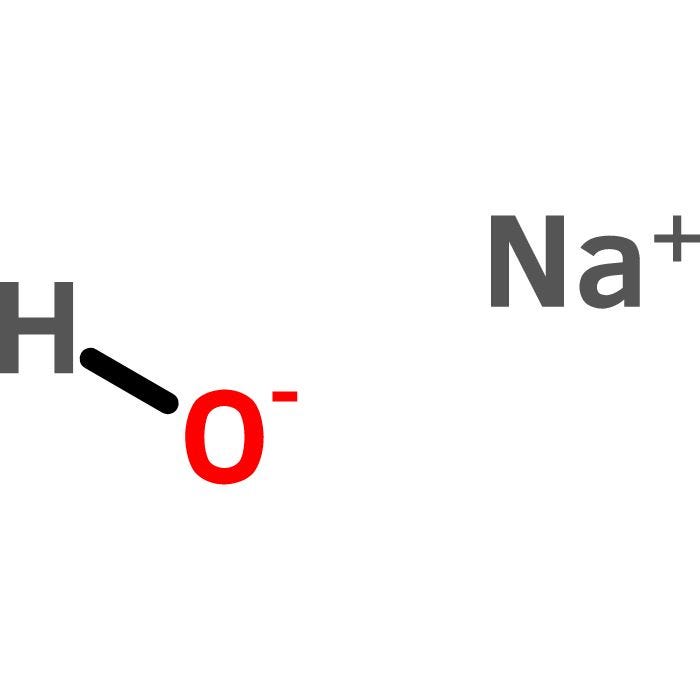
| Reactivity |
Hygroscopic. Much heat is evolved when solid material is dissolved in water. Therefore cold water and caution must be used for this process.
Generates considerable heat when a sodium hydroxide solution is mixed with an acid
Sodium hydroxide solution and octanol + diborane during a work-up of a reaction mixture of oxime and diborane in tetrahyrofuran is very exothermic, a mild explosion being noted on one occassion.
Reactive with water, acids (mineral, non-oxidizing, e.g. hydrochloric, hydrofluoric acid, muriatic acid, phosphoric), acids (mineral, oxidizing e.g. chromic acid, hypochlorous acid, nitric acid, sulfuric acid), acids (organic e.g. acetic acid, benzoic acid, formic acid, methanoic acid, oxalic acid), aldehydes (e.g. acetaldehyde, acrolein, chloral hydrate, foraldehyde), carbamates (e.g. carbanolate, carbofuran), esters (e.g. butyl acetate, ethyl acetate, propyl formate), halogenated organics (dibromoethane, hexachlorobenzene, methyl chloride, trichloroethylene), isocyanates (e.g. methyl isocyanate), ketones (acetone, acetophenone, MEK, MIBK), acid chlorides, strong bases, strong oxidizing agents, strong reducing agents, flammable liquids, powdered metals and metals (i.e aluminum, tin, zinc, hafnium, raney nickel), metals (alkali and alkaline e.g. cesium, potassium, sodium), metal compounds (toxic e.g. berylium, lead acetate, nickel carbonyl, tetraethyl lead), mitrides (e.g. potassium nitride, sodium nitride), nitriles (e.g. acetonitrile, methyl cyanide), nitro compounds (organic e.g. nitrobenzene, nitromethane), acetic anhydride, hydroquinone, chlorohydrin, chlorosulfonic acid, ethylene cyanohydrin, glyoxal, hydrosulfuric acid, oleum, propiolactone, acylonitrile, phorosous pentoxide, chloroethanol, chloroform-methanol, tetrahydroborate, cyanogen azide, 1,2,4,5 tetrachlorobenzene, cinnamaldehyde.
Reacts with formaldehyde hydroxide to yield formic acid, and hydrogen. |